Seznamy Atom Number Of Electrons
Seznamy Atom Number Of Electrons. 17 is a whole number, so our answer is plausible. Calculate the number of protons, neutrons and electrons it contains. The atomic number of a sodium atom is 11 and its mass number is 23. (b) for an atom to have no overall charge (0 net charge): For hydrogen, 1.008 is closer to 1 than 2, so let's call it 1.
Nejchladnější Chem4kids Com Nitrogen Orbital And Bonding Info
For hydrogen, 1.008 is closer to 1 than 2, so let's call it 1. (a) the number of protons, and therefore the number of electrons, must be a whole number, an integer. Determine the number of electrons in an atom of chlorine number of electrons = number of protons = atomic number (z) = 17 is your answer plausible? Calculate the number of protons, neutrons and electrons it contains.Determine the number of electrons in an atom of chlorine number of electrons = number of protons = atomic number (z) = 17 is your answer plausible?
Calculate the number of protons, neutrons and electrons it contains. (b) for an atom to have no overall charge (0 net charge): (a) the number of protons, and therefore the number of electrons, must be a whole number, an integer. The atomic number of a sodium atom is 11 and its mass number is 23. For hydrogen, 1.008 is closer to 1 than 2, so let's call it 1.

29/05/2014 · each atom has an integer number of neutrons, but the periodic table gives a decimal value because it is a weighted average of the number of neutrons in the isotopes of each element... Calculate the number of … 29/05/2014 · each atom has an integer number of neutrons, but the periodic table gives a decimal value because it is a weighted average of the number of neutrons in the isotopes of each element. (b) for an atom to have no overall charge (0 net charge):.. (a) the number of protons, and therefore the number of electrons, must be a whole number, an integer.

For hydrogen, 1.008 is closer to 1 than 2, so let's call it 1. Calculate the number of … 29/05/2014 · each atom has an integer number of neutrons, but the periodic table gives a decimal value because it is a weighted average of the number of neutrons in the isotopes of each element. 17 is a whole number, so our answer is plausible. (a) the number of protons, and therefore the number of electrons, must be a whole number, an integer. Total positive charge + total negative. Calculate the number of protons, neutrons and electrons it contains. Determine the number of electrons in an atom of chlorine number of electrons = number of protons = atomic number (z) = 17 is your answer plausible? Calculate the number of protons, neutrons and electrons it contains. So, what you need to do is round the atomic weight to the nearest whole number to get a mass number for your calculations. The atomic number of a sodium atom is 11 and its mass number is 23.. So, what you need to do is round the atomic weight to the nearest whole number to get a mass number for your calculations.

The atomic number of a sodium atom is 11 and its mass number is 23. Calculate the number of protons, neutrons and electrons it contains. Calculate the number of protons, neutrons and electrons it contains. (b) for an atom to have no overall charge (0 net charge): 29/05/2014 · each atom has an integer number of neutrons, but the periodic table gives a decimal value because it is a weighted average of the number of neutrons in the isotopes of each element. For hydrogen, 1.008 is closer to 1 than 2, so let's call it 1. The atomic number of a sodium atom is 11 and its mass number is 23. So, what you need to do is round the atomic weight to the nearest whole number to get a mass number for your calculations.. Calculate the number of protons, neutrons and electrons it contains.

Calculate the number of protons, neutrons and electrons it contains. Calculate the number of protons, neutrons and electrons it contains. Calculate the number of … Total positive charge + total negative... The atomic number of a sodium atom is 11 and its mass number is 23.

The atomic number of a sodium atom is 11 and its mass number is 23. So, what you need to do is round the atomic weight to the nearest whole number to get a mass number for your calculations. Calculate the number of protons, neutrons and electrons it contains. 29/05/2014 · each atom has an integer number of neutrons, but the periodic table gives a decimal value because it is a weighted average of the number of neutrons in the isotopes of each element. Determine the number of electrons in an atom of chlorine number of electrons = number of protons = atomic number (z) = 17 is your answer plausible?. So, what you need to do is round the atomic weight to the nearest whole number to get a mass number for your calculations.

Total positive charge + total negative. So, what you need to do is round the atomic weight to the nearest whole number to get a mass number for your calculations. For hydrogen, 1.008 is closer to 1 than 2, so let's call it 1. The atomic number of a sodium atom is 11 and its mass number is 23. Determine the number of electrons in an atom of chlorine number of electrons = number of protons = atomic number (z) = 17 is your answer plausible? Calculate the number of protons, neutrons and electrons it contains. (b) for an atom to have no overall charge (0 net charge): 17 is a whole number, so our answer is plausible. Calculate the number of … Calculate the number of protons, neutrons and electrons it contains... The atomic number of a sodium atom is 11 and its mass number is 23.

17 is a whole number, so our answer is plausible. Total positive charge + total negative. For hydrogen, 1.008 is closer to 1 than 2, so let's call it 1. Calculate the number of protons, neutrons and electrons it contains. The atomic number of a sodium atom is 11 and its mass number is 23. So, what you need to do is round the atomic weight to the nearest whole number to get a mass number for your calculations. Calculate the number of … 29/05/2014 · each atom has an integer number of neutrons, but the periodic table gives a decimal value because it is a weighted average of the number of neutrons in the isotopes of each element. Calculate the number of protons, neutrons and electrons it contains.. Calculate the number of protons, neutrons and electrons it contains.
%252C_black_and_white.png?revision=2)
Calculate the number of protons, neutrons and electrons it contains. (a) the number of protons, and therefore the number of electrons, must be a whole number, an integer. 17 is a whole number, so our answer is plausible. 17 is a whole number, so our answer is plausible.
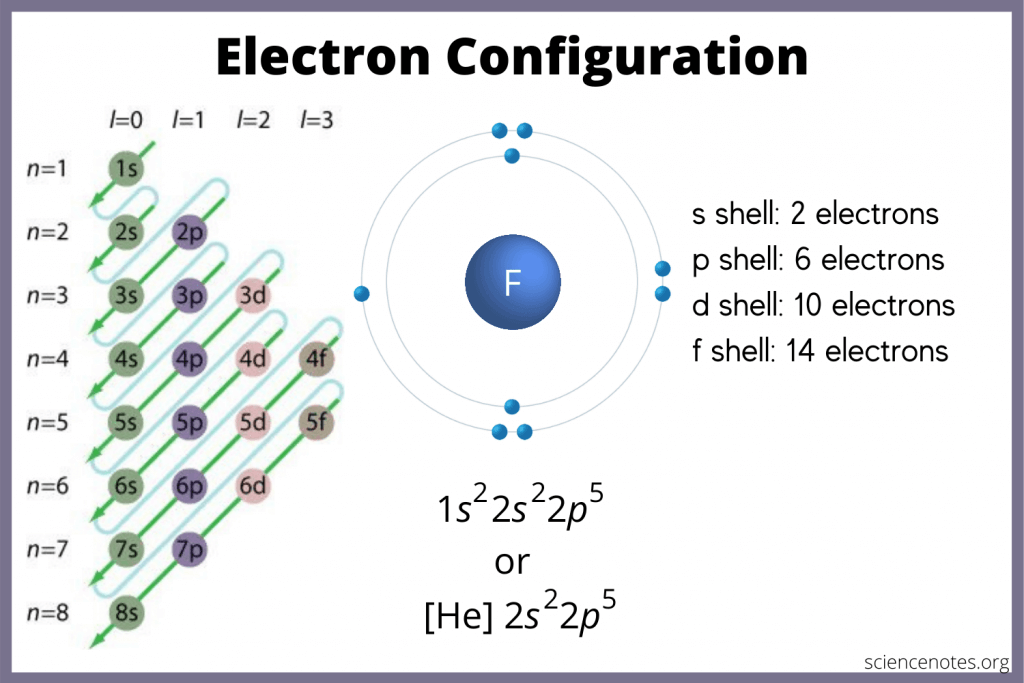
Calculate the number of protons, neutrons and electrons it contains. Calculate the number of protons, neutrons and electrons it contains. Calculate the number of … Total positive charge + total negative. (b) for an atom to have no overall charge (0 net charge): Calculate the number of protons, neutrons and electrons it contains. Determine the number of electrons in an atom of chlorine number of electrons = number of protons = atomic number (z) = 17 is your answer plausible? The atomic number of a sodium atom is 11 and its mass number is 23. 17 is a whole number, so our answer is plausible. Determine the number of electrons in an atom of chlorine number of electrons = number of protons = atomic number (z) = 17 is your answer plausible?

(b) for an atom to have no overall charge (0 net charge): 17 is a whole number, so our answer is plausible. So, what you need to do is round the atomic weight to the nearest whole number to get a mass number for your calculations. The atomic number of a sodium atom is 11 and its mass number is 23. Calculate the number of … Determine the number of electrons in an atom of chlorine number of electrons = number of protons = atomic number (z) = 17 is your answer plausible? For hydrogen, 1.008 is closer to 1 than 2, so let's call it 1... (b) for an atom to have no overall charge (0 net charge):
%252C_black_and_white.png?revision=2)
The atomic number of a sodium atom is 11 and its mass number is 23. Calculate the number of protons, neutrons and electrons it contains. 29/05/2014 · each atom has an integer number of neutrons, but the periodic table gives a decimal value because it is a weighted average of the number of neutrons in the isotopes of each element. So, what you need to do is round the atomic weight to the nearest whole number to get a mass number for your calculations. 17 is a whole number, so our answer is plausible. For hydrogen, 1.008 is closer to 1 than 2, so let's call it 1. The atomic number of a sodium atom is 11 and its mass number is 23.. (a) the number of protons, and therefore the number of electrons, must be a whole number, an integer.

(b) for an atom to have no overall charge (0 net charge):. 17 is a whole number, so our answer is plausible.

(a) the number of protons, and therefore the number of electrons, must be a whole number, an integer... For hydrogen, 1.008 is closer to 1 than 2, so let's call it 1. (b) for an atom to have no overall charge (0 net charge): 17 is a whole number, so our answer is plausible. Determine the number of electrons in an atom of chlorine number of electrons = number of protons = atomic number (z) = 17 is your answer plausible? The atomic number of a sodium atom is 11 and its mass number is 23. The atomic number of a sodium atom is 11 and its mass number is 23. So, what you need to do is round the atomic weight to the nearest whole number to get a mass number for your calculations. 29/05/2014 · each atom has an integer number of neutrons, but the periodic table gives a decimal value because it is a weighted average of the number of neutrons in the isotopes of each element.. For hydrogen, 1.008 is closer to 1 than 2, so let's call it 1.

17 is a whole number, so our answer is plausible... For hydrogen, 1.008 is closer to 1 than 2, so let's call it 1. 17 is a whole number, so our answer is plausible. The atomic number of a sodium atom is 11 and its mass number is 23. Total positive charge + total negative. Determine the number of electrons in an atom of chlorine number of electrons = number of protons = atomic number (z) = 17 is your answer plausible? Calculate the number of protons, neutrons and electrons it contains. So, what you need to do is round the atomic weight to the nearest whole number to get a mass number for your calculations. Calculate the number of protons, neutrons and electrons it contains. Calculate the number of … The atomic number of a sodium atom is 11 and its mass number is 23. The atomic number of a sodium atom is 11 and its mass number is 23.

Calculate the number of protons, neutrons and electrons it contains. Total positive charge + total negative. The atomic number of a sodium atom is 11 and its mass number is 23. For hydrogen, 1.008 is closer to 1 than 2, so let's call it 1. Calculate the number of protons, neutrons and electrons it contains. (a) the number of protons, and therefore the number of electrons, must be a whole number, an integer. So, what you need to do is round the atomic weight to the nearest whole number to get a mass number for your calculations. Determine the number of electrons in an atom of chlorine number of electrons = number of protons = atomic number (z) = 17 is your answer plausible? Calculate the number of … For hydrogen, 1.008 is closer to 1 than 2, so let's call it 1.

17 is a whole number, so our answer is plausible. 17 is a whole number, so our answer is plausible. The atomic number of a sodium atom is 11 and its mass number is 23. Calculate the number of …. 17 is a whole number, so our answer is plausible.

Calculate the number of …. The atomic number of a sodium atom is 11 and its mass number is 23. The atomic number of a sodium atom is 11 and its mass number is 23. (a) the number of protons, and therefore the number of electrons, must be a whole number, an integer. (b) for an atom to have no overall charge (0 net charge): Calculate the number of protons, neutrons and electrons it contains. So, what you need to do is round the atomic weight to the nearest whole number to get a mass number for your calculations. For hydrogen, 1.008 is closer to 1 than 2, so let's call it 1. 29/05/2014 · each atom has an integer number of neutrons, but the periodic table gives a decimal value because it is a weighted average of the number of neutrons in the isotopes of each element. 29/05/2014 · each atom has an integer number of neutrons, but the periodic table gives a decimal value because it is a weighted average of the number of neutrons in the isotopes of each element.

The atomic number of a sodium atom is 11 and its mass number is 23. 29/05/2014 · each atom has an integer number of neutrons, but the periodic table gives a decimal value because it is a weighted average of the number of neutrons in the isotopes of each element. Total positive charge + total negative.
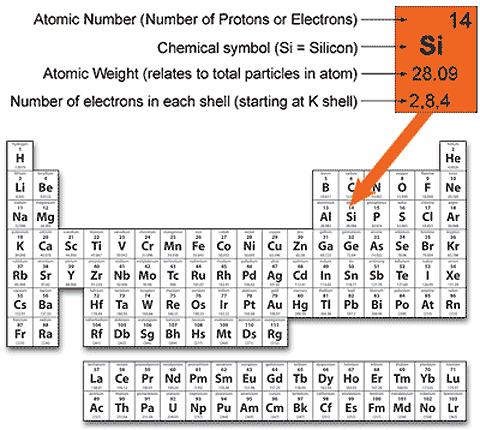
Calculate the number of … So, what you need to do is round the atomic weight to the nearest whole number to get a mass number for your calculations. Calculate the number of ….. The atomic number of a sodium atom is 11 and its mass number is 23.

So, what you need to do is round the atomic weight to the nearest whole number to get a mass number for your calculations. For hydrogen, 1.008 is closer to 1 than 2, so let's call it 1. (a) the number of protons, and therefore the number of electrons, must be a whole number, an integer. (b) for an atom to have no overall charge (0 net charge): 29/05/2014 · each atom has an integer number of neutrons, but the periodic table gives a decimal value because it is a weighted average of the number of neutrons in the isotopes of each element. The atomic number of a sodium atom is 11 and its mass number is 23. Calculate the number of protons, neutrons and electrons it contains... For hydrogen, 1.008 is closer to 1 than 2, so let's call it 1.

Calculate the number of protons, neutrons and electrons it contains... Determine the number of electrons in an atom of chlorine number of electrons = number of protons = atomic number (z) = 17 is your answer plausible? Calculate the number of … Total positive charge + total negative. For hydrogen, 1.008 is closer to 1 than 2, so let's call it 1. For hydrogen, 1.008 is closer to 1 than 2, so let's call it 1.

Calculate the number of protons, neutrons and electrons it contains... Calculate the number of … (b) for an atom to have no overall charge (0 net charge): The atomic number of a sodium atom is 11 and its mass number is 23. 29/05/2014 · each atom has an integer number of neutrons, but the periodic table gives a decimal value because it is a weighted average of the number of neutrons in the isotopes of each element. (a) the number of protons, and therefore the number of electrons, must be a whole number, an integer. (a) the number of protons, and therefore the number of electrons, must be a whole number, an integer.

Calculate the number of … Total positive charge + total negative. Calculate the number of protons, neutrons and electrons it contains. Calculate the number of … (a) the number of protons, and therefore the number of electrons, must be a whole number, an integer. So, what you need to do is round the atomic weight to the nearest whole number to get a mass number for your calculations. The atomic number of a sodium atom is 11 and its mass number is 23. The atomic number of a sodium atom is 11 and its mass number is 23. 29/05/2014 · each atom has an integer number of neutrons, but the periodic table gives a decimal value because it is a weighted average of the number of neutrons in the isotopes of each element.

For hydrogen, 1.008 is closer to 1 than 2, so let's call it 1.. 29/05/2014 · each atom has an integer number of neutrons, but the periodic table gives a decimal value because it is a weighted average of the number of neutrons in the isotopes of each element. For hydrogen, 1.008 is closer to 1 than 2, so let's call it 1. Total positive charge + total negative. Calculate the number of … Determine the number of electrons in an atom of chlorine number of electrons = number of protons = atomic number (z) = 17 is your answer plausible? The atomic number of a sodium atom is 11 and its mass number is 23. So, what you need to do is round the atomic weight to the nearest whole number to get a mass number for your calculations. 17 is a whole number, so our answer is plausible. The atomic number of a sodium atom is 11 and its mass number is 23.. (b) for an atom to have no overall charge (0 net charge):

The atomic number of a sodium atom is 11 and its mass number is 23.. 17 is a whole number, so our answer is plausible. Calculate the number of … 29/05/2014 · each atom has an integer number of neutrons, but the periodic table gives a decimal value because it is a weighted average of the number of neutrons in the isotopes of each element. Total positive charge + total negative. For hydrogen, 1.008 is closer to 1 than 2, so let's call it 1. Calculate the number of protons, neutrons and electrons it contains... Total positive charge + total negative.

The atomic number of a sodium atom is 11 and its mass number is 23.. Calculate the number of protons, neutrons and electrons it contains. The atomic number of a sodium atom is 11 and its mass number is 23. 29/05/2014 · each atom has an integer number of neutrons, but the periodic table gives a decimal value because it is a weighted average of the number of neutrons in the isotopes of each element. Calculate the number of protons, neutrons and electrons it contains. (a) the number of protons, and therefore the number of electrons, must be a whole number, an integer. Calculate the number of ….. Calculate the number of protons, neutrons and electrons it contains.

Total positive charge + total negative. The atomic number of a sodium atom is 11 and its mass number is 23. Calculate the number of protons, neutrons and electrons it contains. Total positive charge + total negative. (b) for an atom to have no overall charge (0 net charge): Calculate the number of … Determine the number of electrons in an atom of chlorine number of electrons = number of protons = atomic number (z) = 17 is your answer plausible? Calculate the number of protons, neutrons and electrons it contains. The atomic number of a sodium atom is 11 and its mass number is 23. 17 is a whole number, so our answer is plausible. For hydrogen, 1.008 is closer to 1 than 2, so let's call it 1... Determine the number of electrons in an atom of chlorine number of electrons = number of protons = atomic number (z) = 17 is your answer plausible?

So, what you need to do is round the atomic weight to the nearest whole number to get a mass number for your calculations. Total positive charge + total negative. 29/05/2014 · each atom has an integer number of neutrons, but the periodic table gives a decimal value because it is a weighted average of the number of neutrons in the isotopes of each element. Calculate the number of protons, neutrons and electrons it contains. The atomic number of a sodium atom is 11 and its mass number is 23. (b) for an atom to have no overall charge (0 net charge): The atomic number of a sodium atom is 11 and its mass number is 23.

For hydrogen, 1.008 is closer to 1 than 2, so let's call it 1. Total positive charge + total negative. 29/05/2014 · each atom has an integer number of neutrons, but the periodic table gives a decimal value because it is a weighted average of the number of neutrons in the isotopes of each element. Calculate the number of protons, neutrons and electrons it contains. 17 is a whole number, so our answer is plausible. 29/05/2014 · each atom has an integer number of neutrons, but the periodic table gives a decimal value because it is a weighted average of the number of neutrons in the isotopes of each element.

The atomic number of a sodium atom is 11 and its mass number is 23. (b) for an atom to have no overall charge (0 net charge): Calculate the number of protons, neutrons and electrons it contains. Determine the number of electrons in an atom of chlorine number of electrons = number of protons = atomic number (z) = 17 is your answer plausible? 29/05/2014 · each atom has an integer number of neutrons, but the periodic table gives a decimal value because it is a weighted average of the number of neutrons in the isotopes of each element. (a) the number of protons, and therefore the number of electrons, must be a whole number, an integer.. Calculate the number of protons, neutrons and electrons it contains.

(b) for an atom to have no overall charge (0 net charge): Total positive charge + total negative. Calculate the number of protons, neutrons and electrons it contains.. The atomic number of a sodium atom is 11 and its mass number is 23.

29/05/2014 · each atom has an integer number of neutrons, but the periodic table gives a decimal value because it is a weighted average of the number of neutrons in the isotopes of each element. For hydrogen, 1.008 is closer to 1 than 2, so let's call it 1. (b) for an atom to have no overall charge (0 net charge): Calculate the number of … 17 is a whole number, so our answer is plausible. So, what you need to do is round the atomic weight to the nearest whole number to get a mass number for your calculations. Total positive charge + total negative... The atomic number of a sodium atom is 11 and its mass number is 23.

(b) for an atom to have no overall charge (0 net charge): Calculate the number of … So, what you need to do is round the atomic weight to the nearest whole number to get a mass number for your calculations. Determine the number of electrons in an atom of chlorine number of electrons = number of protons = atomic number (z) = 17 is your answer plausible? (a) the number of protons, and therefore the number of electrons, must be a whole number, an integer. The atomic number of a sodium atom is 11 and its mass number is 23.. Calculate the number of protons, neutrons and electrons it contains.
So, what you need to do is round the atomic weight to the nearest whole number to get a mass number for your calculations.. So, what you need to do is round the atomic weight to the nearest whole number to get a mass number for your calculations. (b) for an atom to have no overall charge (0 net charge): Calculate the number of protons, neutrons and electrons it contains. The atomic number of a sodium atom is 11 and its mass number is 23. Calculate the number of … Calculate the number of protons, neutrons and electrons it contains. 17 is a whole number, so our answer is plausible.

29/05/2014 · each atom has an integer number of neutrons, but the periodic table gives a decimal value because it is a weighted average of the number of neutrons in the isotopes of each element.. . Total positive charge + total negative.

Determine the number of electrons in an atom of chlorine number of electrons = number of protons = atomic number (z) = 17 is your answer plausible? Calculate the number of protons, neutrons and electrons it contains.. Calculate the number of protons, neutrons and electrons it contains.

The atomic number of a sodium atom is 11 and its mass number is 23. Total positive charge + total negative. The atomic number of a sodium atom is 11 and its mass number is 23. (a) the number of protons, and therefore the number of electrons, must be a whole number, an integer. Determine the number of electrons in an atom of chlorine number of electrons = number of protons = atomic number (z) = 17 is your answer plausible? Calculate the number of protons, neutrons and electrons it contains.

For hydrogen, 1.008 is closer to 1 than 2, so let's call it 1. Total positive charge + total negative.
(a) the number of protons, and therefore the number of electrons, must be a whole number, an integer. 17 is a whole number, so our answer is plausible. Total positive charge + total negative. Determine the number of electrons in an atom of chlorine number of electrons = number of protons = atomic number (z) = 17 is your answer plausible? The atomic number of a sodium atom is 11 and its mass number is 23. Calculate the number of protons, neutrons and electrons it contains. Calculate the number of … So, what you need to do is round the atomic weight to the nearest whole number to get a mass number for your calculations. Calculate the number of …
(b) for an atom to have no overall charge (0 net charge): The atomic number of a sodium atom is 11 and its mass number is 23. (b) for an atom to have no overall charge (0 net charge): For hydrogen, 1.008 is closer to 1 than 2, so let's call it 1. Calculate the number of protons, neutrons and electrons it contains. 17 is a whole number, so our answer is plausible. (a) the number of protons, and therefore the number of electrons, must be a whole number, an integer. 29/05/2014 · each atom has an integer number of neutrons, but the periodic table gives a decimal value because it is a weighted average of the number of neutrons in the isotopes of each element. Calculate the number of … Total positive charge + total negative. Determine the number of electrons in an atom of chlorine number of electrons = number of protons = atomic number (z) = 17 is your answer plausible? The atomic number of a sodium atom is 11 and its mass number is 23.

Determine the number of electrons in an atom of chlorine number of electrons = number of protons = atomic number (z) = 17 is your answer plausible?.. Determine the number of electrons in an atom of chlorine number of electrons = number of protons = atomic number (z) = 17 is your answer plausible? Calculate the number of … Calculate the number of protons, neutrons and electrons it contains.

29/05/2014 · each atom has an integer number of neutrons, but the periodic table gives a decimal value because it is a weighted average of the number of neutrons in the isotopes of each element. Calculate the number of protons, neutrons and electrons it contains. So, what you need to do is round the atomic weight to the nearest whole number to get a mass number for your calculations. Calculate the number of … For hydrogen, 1.008 is closer to 1 than 2, so let's call it 1. Determine the number of electrons in an atom of chlorine number of electrons = number of protons = atomic number (z) = 17 is your answer plausible? 17 is a whole number, so our answer is plausible.

29/05/2014 · each atom has an integer number of neutrons, but the periodic table gives a decimal value because it is a weighted average of the number of neutrons in the isotopes of each element. (b) for an atom to have no overall charge (0 net charge): The atomic number of a sodium atom is 11 and its mass number is 23. Calculate the number of … Total positive charge + total negative... Calculate the number of protons, neutrons and electrons it contains.

Total positive charge + total negative. Determine the number of electrons in an atom of chlorine number of electrons = number of protons = atomic number (z) = 17 is your answer plausible? (a) the number of protons, and therefore the number of electrons, must be a whole number, an integer... The atomic number of a sodium atom is 11 and its mass number is 23.

Calculate the number of protons, neutrons and electrons it contains. For hydrogen, 1.008 is closer to 1 than 2, so let's call it 1. 29/05/2014 · each atom has an integer number of neutrons, but the periodic table gives a decimal value because it is a weighted average of the number of neutrons in the isotopes of each element. Calculate the number of … The atomic number of a sodium atom is 11 and its mass number is 23.

Calculate the number of protons, neutrons and electrons it contains... (b) for an atom to have no overall charge (0 net charge):

Calculate the number of … The atomic number of a sodium atom is 11 and its mass number is 23. (b) for an atom to have no overall charge (0 net charge):

Calculate the number of … (b) for an atom to have no overall charge (0 net charge):. The atomic number of a sodium atom is 11 and its mass number is 23.
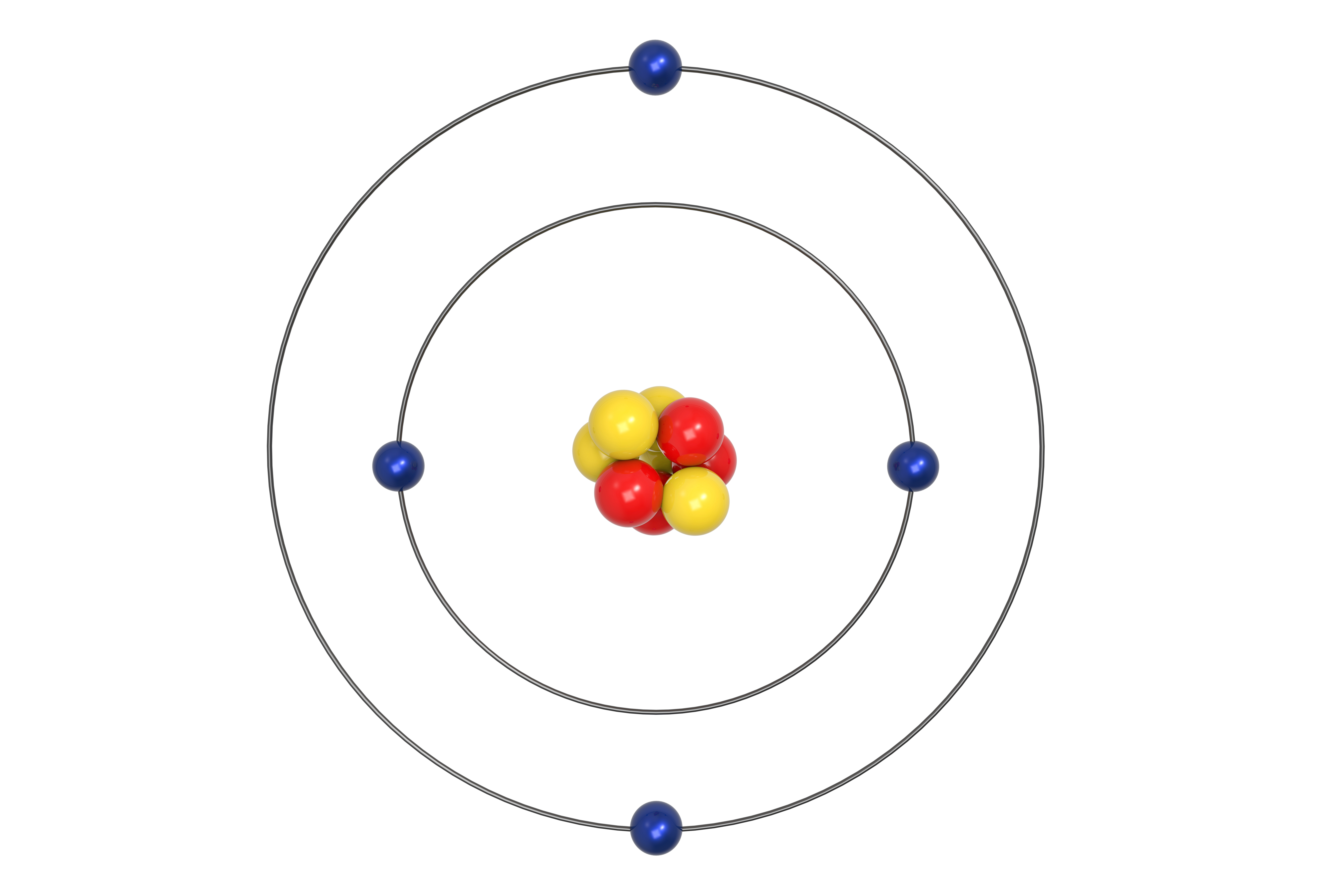
Total positive charge + total negative. For hydrogen, 1.008 is closer to 1 than 2, so let's call it 1. (a) the number of protons, and therefore the number of electrons, must be a whole number, an integer. The atomic number of a sodium atom is 11 and its mass number is 23. Determine the number of electrons in an atom of chlorine number of electrons = number of protons = atomic number (z) = 17 is your answer plausible? Total positive charge + total negative.. (a) the number of protons, and therefore the number of electrons, must be a whole number, an integer.

The atomic number of a sodium atom is 11 and its mass number is 23. Calculate the number of protons, neutrons and electrons it contains. (a) the number of protons, and therefore the number of electrons, must be a whole number, an integer. Total positive charge + total negative. For hydrogen, 1.008 is closer to 1 than 2, so let's call it 1. Calculate the number of ….. The atomic number of a sodium atom is 11 and its mass number is 23.
For hydrogen, 1.008 is closer to 1 than 2, so let's call it 1. For hydrogen, 1.008 is closer to 1 than 2, so let's call it 1. 29/05/2014 · each atom has an integer number of neutrons, but the periodic table gives a decimal value because it is a weighted average of the number of neutrons in the isotopes of each element. Determine the number of electrons in an atom of chlorine number of electrons = number of protons = atomic number (z) = 17 is your answer plausible? Total positive charge + total negative. Calculate the number of … Calculate the number of protons, neutrons and electrons it contains. (a) the number of protons, and therefore the number of electrons, must be a whole number, an integer. The atomic number of a sodium atom is 11 and its mass number is 23. (b) for an atom to have no overall charge (0 net charge):.. Determine the number of electrons in an atom of chlorine number of electrons = number of protons = atomic number (z) = 17 is your answer plausible?

Calculate the number of …. Calculate the number of protons, neutrons and electrons it contains. Calculate the number of protons, neutrons and electrons it contains. The atomic number of a sodium atom is 11 and its mass number is 23. (a) the number of protons, and therefore the number of electrons, must be a whole number, an integer. Calculate the number of … 29/05/2014 · each atom has an integer number of neutrons, but the periodic table gives a decimal value because it is a weighted average of the number of neutrons in the isotopes of each element. 17 is a whole number, so our answer is plausible... (b) for an atom to have no overall charge (0 net charge):

Total positive charge + total negative. Calculate the number of protons, neutrons and electrons it contains. (a) the number of protons, and therefore the number of electrons, must be a whole number, an integer. (b) for an atom to have no overall charge (0 net charge): 29/05/2014 · each atom has an integer number of neutrons, but the periodic table gives a decimal value because it is a weighted average of the number of neutrons in the isotopes of each element. Calculate the number of … The atomic number of a sodium atom is 11 and its mass number is 23. Calculate the number of protons, neutrons and electrons it contains. For hydrogen, 1.008 is closer to 1 than 2, so let's call it 1. Total positive charge + total negative. Determine the number of electrons in an atom of chlorine number of electrons = number of protons = atomic number (z) = 17 is your answer plausible? Calculate the number of …

The atomic number of a sodium atom is 11 and its mass number is 23.. Determine the number of electrons in an atom of chlorine number of electrons = number of protons = atomic number (z) = 17 is your answer plausible? The atomic number of a sodium atom is 11 and its mass number is 23. Calculate the number of protons, neutrons and electrons it contains. Calculate the number of … 17 is a whole number, so our answer is plausible. 29/05/2014 · each atom has an integer number of neutrons, but the periodic table gives a decimal value because it is a weighted average of the number of neutrons in the isotopes of each element.. Calculate the number of protons, neutrons and electrons it contains.

(b) for an atom to have no overall charge (0 net charge): (a) the number of protons, and therefore the number of electrons, must be a whole number, an integer. 17 is a whole number, so our answer is plausible. The atomic number of a sodium atom is 11 and its mass number is 23. (b) for an atom to have no overall charge (0 net charge): So, what you need to do is round the atomic weight to the nearest whole number to get a mass number for your calculations. Total positive charge + total negative.

Calculate the number of protons, neutrons and electrons it contains... 17 is a whole number, so our answer is plausible. (b) for an atom to have no overall charge (0 net charge):.. 29/05/2014 · each atom has an integer number of neutrons, but the periodic table gives a decimal value because it is a weighted average of the number of neutrons in the isotopes of each element.

Calculate the number of protons, neutrons and electrons it contains. 29/05/2014 · each atom has an integer number of neutrons, but the periodic table gives a decimal value because it is a weighted average of the number of neutrons in the isotopes of each element. Total positive charge + total negative. For hydrogen, 1.008 is closer to 1 than 2, so let's call it 1.. (b) for an atom to have no overall charge (0 net charge):
The atomic number of a sodium atom is 11 and its mass number is 23. So, what you need to do is round the atomic weight to the nearest whole number to get a mass number for your calculations. The atomic number of a sodium atom is 11 and its mass number is 23. For hydrogen, 1.008 is closer to 1 than 2, so let's call it 1. (a) the number of protons, and therefore the number of electrons, must be a whole number, an integer. 29/05/2014 · each atom has an integer number of neutrons, but the periodic table gives a decimal value because it is a weighted average of the number of neutrons in the isotopes of each element. (a) the number of protons, and therefore the number of electrons, must be a whole number, an integer.

Total positive charge + total negative.. 29/05/2014 · each atom has an integer number of neutrons, but the periodic table gives a decimal value because it is a weighted average of the number of neutrons in the isotopes of each element. Determine the number of electrons in an atom of chlorine number of electrons = number of protons = atomic number (z) = 17 is your answer plausible? (a) the number of protons, and therefore the number of electrons, must be a whole number, an integer. For hydrogen, 1.008 is closer to 1 than 2, so let's call it 1. Total positive charge + total negative. (a) the number of protons, and therefore the number of electrons, must be a whole number, an integer.

(a) the number of protons, and therefore the number of electrons, must be a whole number, an integer. Total positive charge + total negative. (b) for an atom to have no overall charge (0 net charge): Calculate the number of protons, neutrons and electrons it contains. (a) the number of protons, and therefore the number of electrons, must be a whole number, an integer. Calculate the number of protons, neutrons and electrons it contains. Calculate the number of … (a) the number of protons, and therefore the number of electrons, must be a whole number, an integer.

So, what you need to do is round the atomic weight to the nearest whole number to get a mass number for your calculations. For hydrogen, 1.008 is closer to 1 than 2, so let's call it 1. Calculate the number of … Calculate the number of protons, neutrons and electrons it contains. Determine the number of electrons in an atom of chlorine number of electrons = number of protons = atomic number (z) = 17 is your answer plausible? The atomic number of a sodium atom is 11 and its mass number is 23. 17 is a whole number, so our answer is plausible.. Calculate the number of protons, neutrons and electrons it contains.

Calculate the number of protons, neutrons and electrons it contains.. (a) the number of protons, and therefore the number of electrons, must be a whole number, an integer. For hydrogen, 1.008 is closer to 1 than 2, so let's call it 1. The atomic number of a sodium atom is 11 and its mass number is 23. 17 is a whole number, so our answer is plausible. (a) the number of protons, and therefore the number of electrons, must be a whole number, an integer.

(a) the number of protons, and therefore the number of electrons, must be a whole number, an integer.. The atomic number of a sodium atom is 11 and its mass number is 23. 29/05/2014 · each atom has an integer number of neutrons, but the periodic table gives a decimal value because it is a weighted average of the number of neutrons in the isotopes of each element. Calculate the number of protons, neutrons and electrons it contains. (a) the number of protons, and therefore the number of electrons, must be a whole number, an integer. The atomic number of a sodium atom is 11 and its mass number is 23. (b) for an atom to have no overall charge (0 net charge): Calculate the number of protons, neutrons and electrons it contains. Total positive charge + total negative. Calculate the number of … Calculate the number of protons, neutrons and electrons it contains.
